Cosmic Rays in the Classroom
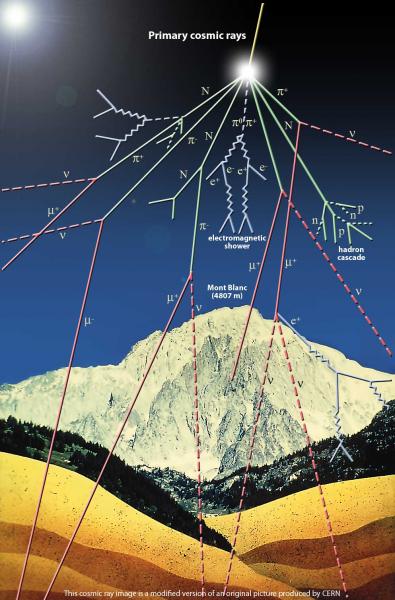
Community 7 membersIn August 1912, Austrian physicist Victor Hess made a historic balloon flight that opened a new window on matter in the universe. As he ascended to 5300 metres, he measured the rate of ionization in the atmosphere and found that it increased to some three times that at sea level. He concluded that penetrating radiation was entering the atmosphere from above. He had discovered cosmic rays.These high-energy particles arriving from outer space are mainly (89%) protons – nuclei of hydrogen, the lightest and most common element in the universe – but they also include nuclei of helium (10%) and heavier nuclei (1%), all the way up to uranium. When they arrive at Earth, they collide with the nuclei of atoms in the upper atmosphere, creating more particles, mainly pions. The charged pions can swiftly decay, emitting particles called muons. Unlike pions, these do not interact strongly with matter, and can travel through the atmosphere to penetrate below ground. The rate of muons arriving at the surface of the Earth is such that about one per second passes through a volume the size of a person’s head.
Where do Cosmic Rays come from? What is their ultimate composition? Can we answer these questions in the school classroom?
In this community of practice, we will explore the science behind cosmic rays in a playful and creative manner.
Students will learn how to build cloud chambers and use them to identify the different components of cosmic radiation.